Written by Mark Jones

Thermosetting polymers cure to form extremely strong bonds, offering resistance to moisture and heat and excellent gap-filling abilities.
A thermoset polymer is formed by a chemical reaction from reactive monomers triggered by the addition of a hardener, UV light, or other triggering event. The properties of the cured polymer are different from the uncured resin and the curing chemistry is irreversible. Once formed, a polymer cannot be returned to the starting point.
In contrast, thermoplastics are polymers that are melted and shaped. This means that, in principle, they can be remelted and reused. With thermosets, you only get one chance.
Thermoset adhesives are thermoset polymers used as adhesives. Unreacted monomers are present in the formulation. The reaction, called curing, converts the monomer formulation into a solid polymer.
Several factors can initiate curing, including:
- An environmental cause, such as water
- The addition of a hardener, which catalyzes the reaction
- A secondary reactive resin component
- A stimulus, such as heat or light
Ultimately, a chemical reaction is responsible for the curing of the adhesive.
Thermoset adhesives rely on a reaction that transforms liquid or gel formulations into solid, reliable adhesives. Typically, two reactive monomers are mixed to start the curing process, supplied as a two-component system. One-part formulations also exist and usually rely on water as part of the cure chemistry. This is the mechanism for some polyurethane adhesives.
One of the most widely available and used thermoset adhesives is epoxies. As with most two-component systems, epoxies have two components, a resin and hardener, mixed in equal parts to initiate the curing process. This combination is applied between surfaces to be bound and clamped. Over time, the mixture hardens to form a strongly bonded joint.
 There’s considerable art in the formulation of thermoset adhesives. One parameter used to adjust this art form is the cure time. Generally, curing takes 10 minutes to a few hours, but it can take several days, depending on the adhesive.
There’s considerable art in the formulation of thermoset adhesives. One parameter used to adjust this art form is the cure time. Generally, curing takes 10 minutes to a few hours, but it can take several days, depending on the adhesive.
Other parameters for mastering the formula are the initial tack and the tendency for a just-mixed adhesive to hold fast. It’s also possible to manipulate the cured resin’s properties with additives, fibers to improve strength, and rubbery materials to make more resilient glue joints. Thermoset adhesives are adaptable for various applications and industries.
Two-component adhesives provide quality bonds, easy storage, and relatively simple processing. One-component systems — which can be liquids, pastes, or gels — can offer processing advantages. They either rely on environmental inputs to initiate curing or contain materials that are triggered by light or increased temperatures. Some formulas must be cold stored to delay curing.
One-part resins are used in pre-impregnated (prepreg) composites, where heating in a mold creates the final shape, with the thermoset adhesive bonding fibers in the composite into a unified part.
Thermoset adhesives offer extremely high strength, excellent gap-filling ability, and resistance to moisture and heat. Examples include epoxy, polyurethane, and polyester resins. The top-performing and most modern adhesives are thermosets and, for this reason, they’re often used in demanding applications.
Six examples of thermoset resins include:
1. Epoxy resin – the most widely used and commonly available thermoset adhesive. Epoxy refers to the epoxide or oxirane ring, which is the reactive part of the resin. Epoxy resins use di-epoxide molecules to react with amine or alcohol functionality in a hardener. The di-epoxide crosslinks the resin forming a strong, crystalline polymer.
They’re used on various substrates, including wood, metal, and many polymers. Cheap epoxy adhesives sold at hardware and home improvement shops are surprisingly versatile. Exotic formulations, requiring unique curing agents and additive packages, find use in aerospace, automotive, and sporting-good applications. Even golf clubs are held together with specially designed epoxy resins
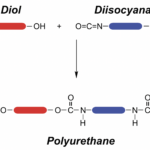
 2. Polyurethane resin – relied on for its high performance. The cure chemistry is due to the presence of isocyanates that react with alcohols or amines. The use of di-isocyanate causes crosslinking, analogous to the function of di-epoxide in epoxy resins. Polyurethanes have high initial tack and form exceedingly strong, resilient bonds. They’re also naturally flexible, offering resilience to bonds not found with other adhesives.
2. Polyurethane resin – relied on for its high performance. The cure chemistry is due to the presence of isocyanates that react with alcohols or amines. The use of di-isocyanate causes crosslinking, analogous to the function of di-epoxide in epoxy resins. Polyurethanes have high initial tack and form exceedingly strong, resilient bonds. They’re also naturally flexible, offering resilience to bonds not found with other adhesives.
3. Silicones – high-performance thermoset adhesives. Various cure chemistries crosslink polysiloxanes into resins that are non-reactive, stable to high temperatures, and resistant to UV degradation. Well-known silicone caulking is a common silicone resin. Exotic silicones find use in electronics, aerospace, automotive, lighting, and other applications.
4. Polyester resin – often used as a lower-cost alternative to epoxy resin for composite structures, especially fiberglass. The cure chemistry is a free-radical chain reaction typically initiated by an organic peroxide. Polyesters do not cure well as thin films, limiting their use as an adhesive for gap-filling applications. The bond strength is lower than epoxy, less resistant to moisture, and it off-gasses volatile organic compounds or VOCs.
5. Phenol formaldehyde resins (PF) – or phenolic resins are one of the earliest forms of plastic. They’re typically used as an adhesive for laminating materials such as paper and wood, and particularly plywood for external use. These are industrial adhesives, rarely applied for individual use.
6. Polyimide adhesives – specialty adhesives that are usually a one-component system that’s cured by heating. Polyimide adhesives offer excellent mechanical properties that make them attractive for use in harsh environments, such as bonding metal aerospace components. These adhesives have high thermal, thermos oxidative, and stable chemical properties and can be used at temperatures up to 500° C (930° F).
Thermoset resins rely on the reactivity of a monomer present in the uncured resin. However, some of these reactive molecules do pose health risks. Be sure to read and follow directions when using thermoset adhesives. Proper personal protective equipment is a must.








Tell Us What You Think!